- Blog
- R2r omnisphere keygen download
- Georgia italic font free download
- Cv in tabular form example
- Fnaf 2 free full game download
- What research has been conducted about scid
- Sims 4 model career 2020
- He developed the atomic theory
- Scoring a moca test
- Logos de among us
- Superior 4l60e shift kit instructions
- Real estate agent daily schedule pdf
- Youtube banner template size
- Horse blanket girl not blurred
- Visual watermark download free
- Jojo the high road album download zip
- Erotic asian massage santa ana ca map
- Cuphead full game free
- Games solitaire klondike free
- Download free internet web Poker software
- Smartgames roadblock game
- Fluidsim 5 license key
- Synergy wellness and medispa
- Google sheet template monthly expenses
- Local moving checklist
- Starcraft remaster skin
- Iphone sim unlock toolkit
- Google maps satellite imagery
- Iridium metallic gmc denali 2018
- Chal jivi laiye gujarati movie online watch
- Moca medical test scoring
- Molar mass from the periodic table
- Terraria free download pc 2021
- Crown of elessar the noble collection
- Unblocked games sports head basketball unblocked
- Microsoft office 2021 professional plus free download
- Bloons tower defense 5 unblocked hacked 66
- Mmpi a online free test
- Online sketchbook online sketchpad
- Download sims 4 wicked whims mod torrent
- Sonic the mania game
- Sims 2 pets expansion pack free download
- Terraria ios free pc download
- Random house sigil generator
- Mp3 youtube music download free
- Molar masses periodic table
- Website banner design templates free
- Decibel rating chart
- Idm crack with serial number free download
The eyes were retained by the Literary & Philosophical Society and donated to the museum in 1997. 'Perfectly colourless' was the result, proving his theory to be incorrect.ĭNA analysis carried out in 1995 and published in the journal Science, 150 years after his death, revealed that Dalton lacked the gene for the receptor sensitive to medium wavelength (green) light, and in fact suffered from deuteranopia, or red-green colour blindness-a condition still referred to as Daltonism. On 28 July 1844, the day after he died, local doctor Joseph Ransome performed the autopsy. He suggested that the colour of the fluid in the eyes, known as the vitreous humour, acted as a filter to certain colours in the spectrum.ĭalton’s ideas were met with resistance from some of his contemporaries at the time, so to test his theory, Dalton donated his eyes for examination after death. John Dalton, English meteorologist and chemist, a pioneer in the development of modern atomic theory. In addition to his work with atoms, Dalton also developed a theory to explain colour vision deficiency (or colour blindness), from which he himself suffered. He was also a figurehead in the world of meteorology. Below are the four principles or claims that compose Democritus atomic theory: 1) All things are composed of the atomos or the fundamental particles, atoms. Democritus developed a theory in which he held that all matter is made up of tiny, indivisible particles that he called atoms that interact with other atoms mechanically. This is because his work hugely influenced the development of the atomic model as we know it today. Robert Oppenheimer’s life.While Oppenheimer will always be recognized as the father of the atomic bomb, his early contributions to quantum mechanics form the bedrock of modern quantum chemistry. Democritus is widely considered to be the father of modern atomic theory. Through his experimentation, Dalton not only formulated a new atomic theory to explain chemical reactions, upon which much of modern chemistry and physics is based, but he also developed a theory to explain colour vision deficiency, from which he himself suffered. The release of the film Oppenheimer, in July 2023, has renewed interest in the enigmatic scientist J. He read over 100 papers to the Society, and became its Secretary, Vice-President and, ultimately, President. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. He proposed a nuclear model where electrons orbit a central, positively charged nucleus, drastically altering the understanding of atomic structure. Though sometimes criticised for the quality of his experiments, Dalton was an enthusiastic investigator who worked late most evenings. Bohr model, description of the structure of atoms, especially that of hydrogen, proposed (1913) by the Danish physicist Niels Bohr. Ernest Rutherford (1871 1937) Rutherford’s gold foil experiment led to the discovery of the atomic nucleus. Through this, he gained access to a well-equipped research laboratory, where his scientific output flourished.

The Society gave him a room for teaching and research at its premises on George Street.

It was a discussion group set up to share scientific ideas at a time when science had yet to become a profession. Attempts to trace precisely how Dalton developed this theory have proved futile even Dalton’s own recollections on the subject are incomplete.
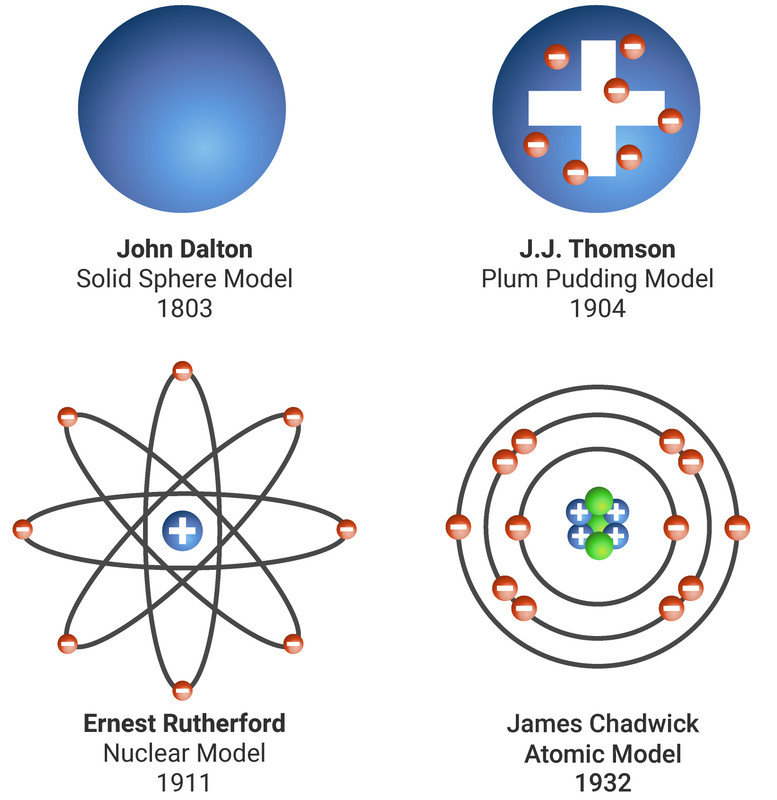
See all videos for this article By far Dalton’s most influential work in chemistry was his atomic theory.

\)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles.Soon after moving to Manchester, Dalton joined the Literary & Philosophical Society, which was at the centre of the city's scientific and business community. John Dalton and the development of the atomic theory.
- Blog
- R2r omnisphere keygen download
- Georgia italic font free download
- Cv in tabular form example
- Fnaf 2 free full game download
- What research has been conducted about scid
- Sims 4 model career 2020
- He developed the atomic theory
- Scoring a moca test
- Logos de among us
- Superior 4l60e shift kit instructions
- Real estate agent daily schedule pdf
- Youtube banner template size
- Horse blanket girl not blurred
- Visual watermark download free
- Jojo the high road album download zip
- Erotic asian massage santa ana ca map
- Cuphead full game free
- Games solitaire klondike free
- Download free internet web Poker software
- Smartgames roadblock game
- Fluidsim 5 license key
- Synergy wellness and medispa
- Google sheet template monthly expenses
- Local moving checklist
- Starcraft remaster skin
- Iphone sim unlock toolkit
- Google maps satellite imagery
- Iridium metallic gmc denali 2018
- Chal jivi laiye gujarati movie online watch
- Moca medical test scoring
- Molar mass from the periodic table
- Terraria free download pc 2021
- Crown of elessar the noble collection
- Unblocked games sports head basketball unblocked
- Microsoft office 2021 professional plus free download
- Bloons tower defense 5 unblocked hacked 66
- Mmpi a online free test
- Online sketchbook online sketchpad
- Download sims 4 wicked whims mod torrent
- Sonic the mania game
- Sims 2 pets expansion pack free download
- Terraria ios free pc download
- Random house sigil generator
- Mp3 youtube music download free
- Molar masses periodic table
- Website banner design templates free
- Decibel rating chart
- Idm crack with serial number free download
